There are several pathways that alone or together influence the development of ADHD.
The first pathway is genetic predisposition, in which a combination of different inherited gene variants usually leads to an imbalance in the brain. Depending on which processes in the brain are affected, different ADHD symptoms develop. Although rare, there are also monogenetic causes of ADHD, in which the disorder of a single gene causes ADHD symptoms. Genetic predisposition is the most common pathway for the development of ADHD and accounts for around 76% of causes.
The second pathway for the development of ADHD is the environment. There are a variety of environmental factors that can promote ADHD prenatally, during birth, in childhood (and more rarely afterwards). These include toxins or medication, chronic severe stress and illnesses such as encephalitis, thyroid problems and many others.
The third pathway is epigenetics, in which environmental influences can be passed on to offspring. Epigenetic influences change gene expression and thus the amount of proteins produced that are encoded by the gene. This can affect neurotransmitter levels in the brain
In epigenetics, however, only the epigenetic changes in gene expression are passed on, not the actual gene variants themselves. Epigenetic influences are therefore only passed on over a few generations.
Finally, there are interactions between genes and the environment that influence the development of ADHD. Some gene variants are more sensitive to environmental influences and can lead to a positive or negative change if they are exposed or encouraged accordingly. The relationship between genetic influences and environmental influences on an existing ADHD can change over a person’s lifetime.
1. Pathways of ADHD development¶
1.1. Genes as the cause of ADHD¶
1.1.1. Interaction of several gene variants¶
ADHD can result from a random coincidence of a sum of inherited gene variants, without the need for environmental influences. Let’s call it “genetically inherited ADHD”.
If a sufficient number of inherited gene variants act in a similar direction, their influences can add up to create an imbalance in the brain that can no longer be corrected by the brain’s many mutually balancing regulatory circuits. Processes can then be disrupted. Depending on which processes are affected (e.g. which neurotransmitters are increased or decreased in which regions of the brain), different disorders can arise.
If several gene variants, each of which has only a small effect on a reduced dopamine and noradrenaline effect in the brain, together reduce this effect too much, the typical ADHD symptoms can develop.
To date, many hundreds of genes relevant to ADHD are known or suspected. ⇒ Candidate genes for ADHD
Genetic causation is the most common pathway. At 75 to 80 %, the genetic component of ADHD is somewhat stronger than that for intelligence. However, these values were mainly obtained from twin studies, which are only of limited significance due to similar living environments. Genetic studies have so far only been able to explain around 20 to 30 % of the development of ADHD genetically.
A few other inheritance rates for comparison:
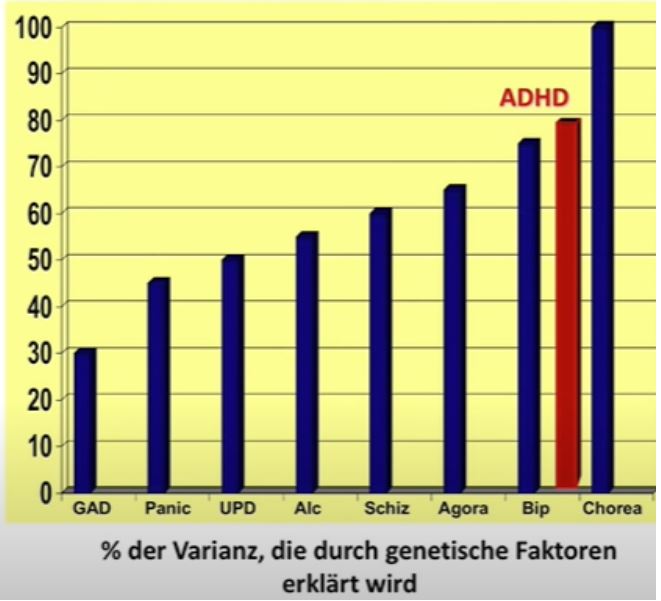
Source: © 2017 www.dasGehirn.info; Lecture Andreas Reif: Focus on ADHD, minute 14.
Generalized anxiety disorder: 30 %
Panic disorder: 45 %
Depression: 50 %
Alcohol dependence: 55 %
Schizophrenia: 60 %
Agoraphobia: 65 %
Bipolar Disorder: 75 %
Huntington’s disease: 100 %
Heritability means the proportion of genes in the causes of the disorder, but not that x% of the children of people with ADHD develop the respective disorder.
1.1.2. Monogenetic causes¶
Rare genetic neurodevelopmental disorders (RGNDs) affect up to 3% of the population [1]. RGNDs are often associated with intellectual disability and psychiatric comorbidities such as ASD and ADHD
A collection of genes in which a gene variant alone of the affected gene causes a very high prevalence of ADHD (up to over 80% and more) can be found at Monogenetic causes of ADHD
1.2. Environment as a cause of ADHD¶
Environmental factors can contribute to the development of ADHD. Let’s call it “acquired ADHD”.
1.2.1. Early or chronic severe stress¶
Early or chronic severe stress can permanently change the expression of genes, i.e. their activity. This can trigger the changes in neurotransmitter levels typical of ADHD or alter the effect of neurotransmitters. Stress medicine already describes these mechanisms for depression. According to this “vulnerability-stress model”, most mental disorders arise: Anxiety, Depression, Compulsions, Autism Spectrum Disorders, Borderline PS and many others.
Early severe stressors in terms of child maltreatment sets 45% of the risks of psychiatric disorders beginning in childhood. Severe early stressors in the sense of child maltreatment can be
- Emotional abuse
- Physical abuse
- Sexual abuse
- Living with a drug addict
- Living with a mentally ill family member
- Witness to violent treatment of the mother
- Household member in prison
- Separation or divorce of parents
- Emotional neglect
- Physical neglect
People with ADHD who experienced 5 or more of these stressors had a greatly increased risk of being prescribed psychotropic drugs
-
Anxiolytics: 2.1-fold
- Antidepressants: 2.9-fold
- Antipsychotics: 10.3-fold
- Mood stabilizers: 17.3-fold
People with ADHD who experienced 6 or more of these stressors had a 20-year lower life expectancy.
Early childhood stress experiences are particularly significant because the stress systems in the brain are only just forming in the first few years of life. Early childhood stress causes permanent damage to the physiological stress systems (HPA axis, autonomic nervous system, PFC). The second particularly dangerous phase for ADHD is middle adolescence. People with ADHD who experienced many severe stressors in adolescence were much more likely to retain their ADHD in adulthood than children with few stressors in adolescence.
What is specific to ADHD is that there is a wide range of symptoms that are also typical of acute or chronic stress (whereby they are functional, i.e. serve to cope with the stressor), while in a number of other mental disorders individual stress symptoms are extremely (and therefore dysfunctional).
⇒ Gene disposition plus early childhood stress as a cause of other mental disorders ⇒ Environmental factors as a cause of ADHD
Acute stress increases the levels of the neurotransmitters dopamine, noradrenaline and serotonin as well as the hormones CRH, ACTH and cortisol. Chronic stress reduces dopamine and noradrenaline. If a stress-induced change in neurotransmitter or hormone levels occurs repeatedly, acutely or permanently, in an (early) childhood phase in which the brain systems that work with these neurotransmitters or hormones are growing, the affected brain regions are disturbed in their development. Brain regions that rely on the above-mentioned neurotransmitters or hormones are therefore particularly vulnerable to stress during their growth phases. Many environmental causes that are known to be risk factors for ADHD are known to attack the dopamine and/or noradrenaline system.
If a corresponding genetic disposition to a dopamine / noradrenaline (action) deficit now coincides with environmental causes that in turn cause a dopamine and / or noradrenaline deficiency, such as early or prolonged stress in children, the effects add up and ADHD becomes more likely than if only one of the two causes occurs alone
⇒ ADHD as a chronic stress regulation disorder
⇒ Stress damage - effects of early childhood and/or prolonged stress
The standard works of stress medicine today discuss which areas of the brain develop at which age and deduce from this at which childhood age stress has taken place because the brain regions that are developing during this time are “misaligned”. For example, severe anxiety in the mother during pregnancy only increases the risk of ADHD in certain weeks of pregnancy.
In contrast, stress outside the years of brain development (i.e. from around the age of 25) is less dangerous because it causes less irreversible damage - although it is still toxic.
This makes it clear why a parenting license would make sense. Not as an admission requirement for having children, but as a minimum level of knowledge about what a defenseless being entrusted to its parents needs. This probably won’t help all children. But that is not the point. Seat belts do not protect life in every accident. Every single child whose soul could be saved from injury would be worth it.
⇒ ADHD - Prevention and screening - What parents can do and⇒ Secure attachment beats genetic disposition in ADHD
1.2.2. Diseases¶
Diseases can also trigger ADHD.
For example, encephalitis destroys the dopaminergic cells in the brain and can thus trigger a dopamine deficit that causes symptoms typical of ADHD. As the symptoms are permanent, encephalitis can be considered a possible cause of ADHD. Parkinson’s disease, in which dopaminergic cells also die, as well as other disorders in which the dopaminergic system is affected in the direction of an (active) deficiency, such as schizophrenia or ASD, all have a significantly increased risk of ADHD.
Thyroid problems can also trigger symptoms similar to those of ADHD. As these symptoms can be remedied by adjusting the thyroid hormones, this is not ADHD. The same applies to vitamin or mineral deficiencies, food intolerances and a number of other disorders.
⇒ Nutrition and diet for ADHD
In order to determine the cause of the symptoms and rule out other causes of the disorder, an appropriate differential diagnosis should be carried out when diagnosing ADHD.
⇒ Differential diagnosis of ADHD
1.2.3. Immune system¶
Diseases that stimulate the immune system to produce behavioral changes typical of ADHD are also conceivable. However, there is little evidence for this. It is more likely that illnesses contribute to ADHD by activating the stress systems that mediate most ADHD symptoms - just like psychological stress.
⇒ Immune system and behavior
1.2.4. Other environmental factors¶
A large number of studies have found a considerable number of environmental factors that can favor ADHD, such as toxins or medication (especially during pregnancy or in the first years of life), particularly young or uneducated parents, living environments with little green growth and many more.
⇒ Environmental factors as a cause of ADHD
1.3. Epigenetics (inherited environmental influences and experiences) as the cause of ADHD¶
ADHD acquired through environmental influences can be passed on to one’s own descendants. In contrast to inherited ADHD of the first variant, only the epigenetic changes in gene activity are passed on. Animal experiments have demonstrated the inheritance of such acquired gene expressions over 2 to 4 generations. Let’s call it “epigenetically inherited ADHD”. This cause of ADHD is particularly amenable to prevention through caring, warm parental behavior.
The epigenetic changes are easier to trigger if there is a corresponding disposition and more difficult if there is no disposition. Without a genetic disposition, ADHD is only caused (and can then be passed on) by significantly more intense early childhood stress (of traumatizing intensity: physical abuse, psychological abuse, sexual abuse). More on this under the keyword opportunity/risk genes.
1.4. Interaction of several pathways in the development of ADHD¶
A relevant developmental variant is when an early childhood stress experience is added to an existing genetic predisposition. In this case, the genes responsible for ADHD change epigenetically in such a way that ADHD now manifests itself (⇒ Genetic and epigenetic causes of ADHD - Introduction). Epigenetics means that (intensive) experiences in life can cause changes in gene expression. These acquired changes can in turn be passed on.
2. ADHD: dopamine and noradrenaline deficiency¶
ADHD is characterized in particular by reduced dopamine and noradrenaline levels in the striatum, PFC and cerebellum areas of the brain. While acute stress shows increased dopamine and noradrenaline levels in these areas of the brain, chronic stress, like ADHD, is typically associated with decreased dopamine and noradrenaline levels in these brain regions. Acute and chronic stress show almost identical symptoms to ADHD, as the dysfunctions of the relevant brain regions that trigger the stress/ADHD symptoms occur with every deviation from an average dopamine or noradrenaline level (inverted-U effect).
Stress has a special significance in ADHD.
Stress is a factor that can trigger the development of ADHD, whereby ADHD acquired in this way is hereditary as a disposition.
In addition, ADHD symptoms are largely mediated by the stress systems.
ADHD also makes people with ADHD more sensitive to stress.
These complex interactions have barely been considered in the ADHD literature to date. This makes a comprehensive description of the stress systems and their influences all the more important to us.
⇒ Stress
3. How significant is which pathway of ADHD development?¶
3.1. Genetic component: approx. 76 %¶
ADHD has a strong genetic component of around 76%. This was determined in studies on identical and fraternal twins. The heritability of ADHD is therefore greater than that of intelligence.
This 76% includes the entirety of genetic transmission through a. gene variants and b. environmental influences in the previous 2 to 4 generations, which are passed on through epigenetics. The remaining 24 % are therefore only those environmental influences that affect the people with ADHD themselves.
Up to 90 % of ADHD cases with clinical intensity are genetically caused. However, individual genes are not the cause, even if certain gene variants are more frequently involved. Hundreds of candidate genes are known. It would not be surprising if there were more than 1000. We have more than 150 of them at ⇒ Candidate genes in ADHD Mentioned. Nevertheless, to date only 5% of genetic heritability can be attributed to specific gene variants. This is probably also due to the fact that genes (in most mental disorders) only represent a disposition and do not yet mean that ADHD exists.
Changes in the functioning of the HPA axis (stress axis) also exist in a purely genetic pathway of ADHD.
⇒ Research topic: ADHD in animal models
Heritability should generally be measured within 1 generation. Environmental causes can influence the affected individual and, via epigenetics, 2-4 generations of their offspring. We therefore assume that the proportion of environmental causes of ADHD is higher than 24%. We estimate it to be around 40 %
3.2. Influence of the environment depending on time¶
Interestingly, the relationship between the influence of genes and environment on existing ADHD changes over time.
The degree of hyperactivity/impulsivity and inattention was evaluated in 602 pairs of Chinese twins at the ages of 12, 13 and 15.
Both symptoms were
- The genetic influences
- Were initially moderate and not significant
- Have become substantial and significant over time
- The environmental influences
- Were initially substantial and significant
- Became moderate and insignificant over time.
Throughout youth, the influences resulted from
- The same genes and
- Other, newly emerging environmental factors
In early adolescence, inattention was predominantly kept stable by genetic influences, hyperactivity/impulsivity increasingly by environmental influences.
The genetic influences of ADHD-HI tended to be lower, while the common environmental influences tended to be greater in Chinese than in Western populations.
At least several of the genes relevant to ADHD are only epigenetically activated by early childhood stress in such a way that they contribute to ADHD. A warm, secure, caring attachment style of parents can reduce the risk of epigenetic activation of genes and thus the risk of ADHD. ⇒ ADHD - Prevention and screening - What parents can do
3.3. Gene-environment interactions¶
One study found that environmental stressors modulate genetic risk for ADHD.
In contrast, no statistically significant interactions were found between the Polygenic Risc Score and environmental influences for depression or anxiety symptoms.
The authors recommend that, particularly with regard to children with a family history of ADHD, it should be investigated whether reducing environmental risks can prevent the development of ADHD symptoms.
3.4. Opportunity-risk genes increase environmental influence¶
Some genes are not only risk genes, but can also become a particularly beneficial and strengthening source if required. (Opportunity-risk genes, see ⇒ Attachment style of the parents to the child is particularly important for opportunity/risk genes)
Certain gene variants make their carriers particularly sensitive (high sensitivity). This leads to a particular susceptibility to support (opportunity) but also to the risk of even greater vulnerability. In these people (approx. 15 %), a stress intensity significantly below that of a trauma is sufficient to activate the genetic predisposition to ADHD: even an insecure attachment style (cool, distant, purely performance-oriented, strict, no reliable attachment, etc.) can be sufficient to manifest ADHD epigenetically with the genetic disposition present here.
Without early childhood stress and appropriate support, highly sensitive people are even more stress-resistant than others.
Regardless of a genetic disposition, stress generally makes us more sensitive and susceptible.
The (epi-)gene changes (inherited or developed through their own traumas) are passed on by the people with ADHD to their children “dormant” - traumas can be inherited over several generations. In the children of people with ADHD, activation of the “dormant” gene disposition leads to the development of ADHD.
As the parents have a relevant probability of having the genes that they pass on as a single parent themselves, the parents (or one parent) often also suffer from ADHD. The ADHD symptoms of (untreated) affected parents then make it highly probable that their children are exposed to increased stress and that the inherited gene disposition is thus activated.
Origin

